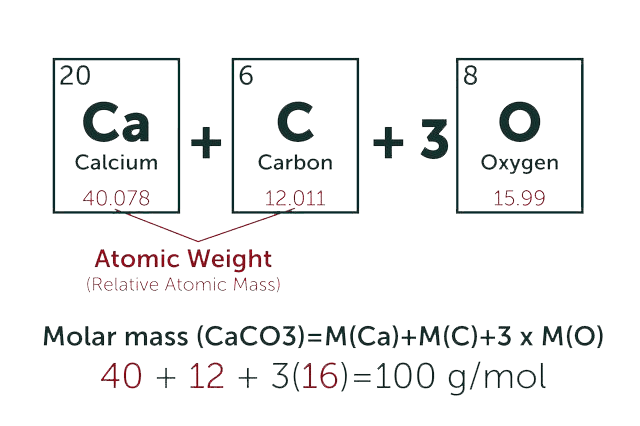Biphenomycin B
* Please be kindly noted products are not for therapeutic use. We do not sell to patients.
| Category | Antibiotics |
| Catalog number | BBF-00151 |
| CAS | 100217-74-1 |
| Molecular Weight | 472.49 |
| Molecular Formula | C23H28N4O7 |
Online Inquiry
Description
Biphenomycin is an antibiotic produced by Streptomyces griseorubiginosus No. 43798. Biphenomycin B has activity against gram-positive bacteria.
Specification
| IUPAC Name | (8S,11S,14S)-14-amino-11-[(2R)-3-amino-2-hydroxypropyl]-5,17-dihydroxy-10,13-dioxo-9,12-diazatricyclo[14.3.1.12,6]henicosa-1(20),2(21),3,5,16,18-hexaene-8-carboxylic acid |
| Canonical SMILES | C1C(C(=O)NC(C(=O)NC(CC2=C(C=CC(=C2)C3=CC1=C(C=C3)O)O)C(=O)O)CC(CN)O)N |
| InChI | InChI=1S/C23H28N4O7/c24-10-15(28)9-17-22(32)27-18(23(33)34)8-14-6-12(2-4-20(14)30)11-1-3-19(29)13(5-11)7-16(25)21(31)26-17/h1-6,15-18,28-30H,7-10,24-25H2,(H,26,31)(H,27,32)(H,33,34)/t15-,16+,17+,18+/m1/s1 |
| InChI Key | OXLPMCIPYGNLJD-OWSLCNJRSA-N |
Properties
| Appearance | Colorless needle Crystal |
| Antibiotic Activity Spectrum | Gram-positive bacteria; Gram-negative bacteria |
| Boiling Point | 953.3°C at 760 mmHg |
| Melting Point | 206-209°C(dec.) |
| Density | 1.359 g/cm3 |
| Solubility | Soluble in water, methanol |
Reference Reading
1. Flexible total synthesis of biphenomycin B
Herbert Waldmann, Yu-Peng He, Hao Tan, Lars Arve, Hans-Dieter Arndt Chem Commun (Camb). 2008 Nov 21;(43):5562-4. doi: 10.1039/b811583d. Epub 2008 Sep 25.
A total synthesis of the biaryl antibiotic biphenomycin B is reported which makes use of three independent building blocks (key steps were a clean Suzuki-Miyaura coupling of a free acid iodide, a novel 4-hydroxyornithine synthesis, and a high-yielding macrolactamization); a practical deprotection protocol allowed isolation of the target compound with excellent recovery and purity.
2. Microwave-assisted intramolecular Suzuki-Miyaura reaction to macrocycle, a concise asymmetric total synthesis of biphenomycin B
Renaud Lépine, Jieping Zhu Org Lett. 2005 Jul 7;7(14):2981-4. doi: 10.1021/ol050949w.
[reaction: see text] A concise and efficient total synthesis of biphenomycin B has been accomplished featuring a key microwave-assisted intramolecular Suzuki-Miyaura reaction for formation of the 15-membered meta,meta-cyclophane 20.
3. Biphenomycin B and derivatives: total synthesis and translation inhibition
Yu-Peng He, Hao Tan, Lars Arve, Sascha Baumann, Herbert Waldmann, Hans-Dieter Arndt Chem Asian J. 2011 Jun 6;6(6):1546-56. doi: 10.1002/asia.201000908. Epub 2011 May 4.
A full account on the synthesis of the antibiotic natural product biphenomycin B and several derivatives is reported, which employs a Suzuki coupling reaction of a free carboxylic acid and macrolactam formation as key transformations. Liberal exchange of the central amino acid was demonstrated. This procedure gave derivatives to study the influence of the polar side chain of the central amino acids on translation inhibition.
Recommended Products
| BBF-03753 | Baicalin | Inquiry |
| BBF-01829 | Deoxynojirimycin | Inquiry |
| BBF-05862 | Epirubicin | Inquiry |
| BBF-01826 | Deoxymannojirimycin | Inquiry |
| BBF-00677 | 3-Amino-3-deoxy-D-glucose | Inquiry |
| BBF-02642 | Lactonamycin | Inquiry |
Bio Calculators
* Our calculator is based on the following equation:
Concentration (start) x Volume (start) = Concentration (final) x Volume (final)
It is commonly abbreviated as: C1V1 = C2V2
* Total Molecular Weight:
g/mol
Tip: Chemical formula is case sensitive. C22H30N4O √ c22h30n40 ╳