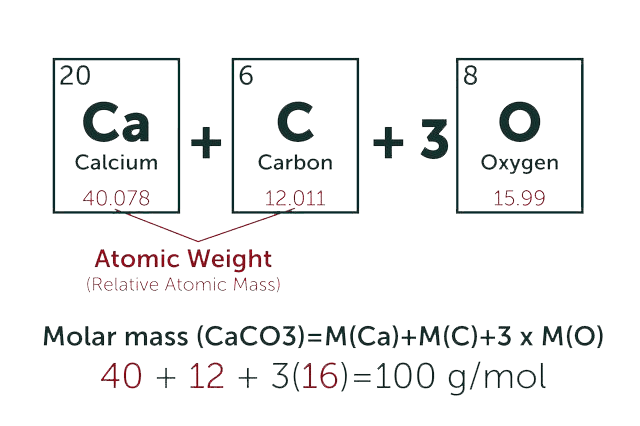1.Electrochemical Behavior and Square Wave Voltammetric Determination of Doxorubicin Hydrochloride
Younghee Hahn and Ho Young Lee. Arch Pharm Res Vol 27, No 1, 31-34, 2004
The detecting system connected to CE was a carbon disk working electrode with an applied potential of 0.95 V vs. a Ag/AgCI (3 M KCI), which measured anodic currents due to the oxidation of two phenolic hydroxyls in the aglycone of daunorubicin (Hu et aL, 2000). Meanwhile, reductive detecting system measured at -0.30 V was preferred to oxidation in which the chromatographic profile suffered severe interference from substances that result from the biological matrix (Ricciarello et aL, 1998). Electrochemical assay often offers selectivity and sensitivity due to the selective detection of electroactive species among the complex samples. The chemical structure of doxorubicin contains a electrochemically a reducible quinone moiety in the aglycone which prompted us to study its electro-chemical behavior by using mercury electrodes, followed by developing the fast and sensitive square wave voltammetric (SWV) procedure for the determination of doxorubicin hydrochloride in the present study.
2.Facile fabrication of thermally responsive Pluronic F127-based nanocapsules for controlled release of doxorubicin hydrochloride
Zhipeng Zeng & Zhiping Peng & Lei Chen & Yiwang Chen. Colloid Polym Sci (2014) 292:1521–1530
The short cross-linking reaction time led to the formation of nanocapsules with thin and low-cross-linked PL shell because the cross-linking reaction between the –NPC groups and –NH2 groups was slow. The difference of the contrast between the PL shell and the hollow core resulted in the typical empty core–shell structure which can be found in the TEM images. After reacting for 20 h, the diameter of the nanocapsules increased to about 200 nm as shown in Fig. 2b. Assuming that the size change of the inner cavity was negligible, the thickness of the PL shell can be estimated to be more than 60 nm. The denser and tighter (with high cross-linking density for long reaction time) PL shell resulted in the empty core–shell structure invisible in the TEM image even though the hollow inner cavity was still there. For the Pluronic F127/HA nanocapsules as shown in Fig. 2d, the smaller and instable nanocapsules were formed because of the slower reaction rate between –COOH groups and –NH2 groups. After reacting for 20 h, the diameter of the Pluronic F127/HA nanocapsules was determined to be about 220 nm. The size of the Pluronic F127/HA nanocapsules was larger than the Pluronic F127/PL nanocapsules because the content of HA was more than PL taken up by the nanocapsules. The average size was consistent with those determined from DLS results.
3.A Method for Evaluation of Therapeutic Dose of Doxorubicin Hydrochloride Using Breast Tumor Cell Culture MCF-7
Ya. D. Shanskij, Yu. A. Ershov*, and V. M. Pechennikov*. Bulletin of Experimental Biology and Medicine, Vol. 148, No. 3, 2009
Statistical data suggest that the use of anticancer drug does not signifi cantly increase the total survival of patient. This is largely associated with difficulties in rational regimen of drug usage. The choice of the dose of anticancer drugs is mainly empirical. This is also true for doxorubicin hydrochloride, antitumor antibiotic of the anthracycline family used for breast cancer management.