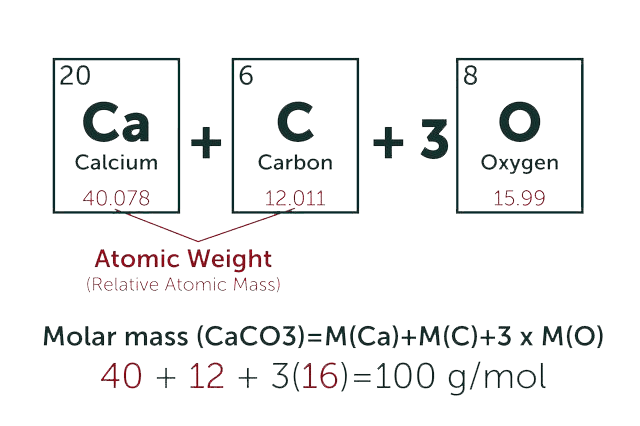Glykenin IV
* Please be kindly noted products are not for therapeutic use. We do not sell to patients.

| Category | Antibiotics |
| Catalog number | BBF-01799 |
| CAS | 134479-76-8 |
| Molecular Weight | 971.13 |
| Molecular Formula | C46H82O21 |
| Purity | 95% |
Online Inquiry
Description
It is produced by the strain of Basidiomycetes sp. It is a glycoside antibiotic, and has anti-gram-positive bacterial activity.
Specification
| Synonyms | GK-Ⅳ; (2S-(2R*,16S*,17R*,21S*))-21-((O-6-O-Acetyl-beta-D-glucopyranosyl-(1-2)-O-beta-D-xylopyranosyl-(1-2)-4-O-acetyl-beta-D-xylopyranosyl)oxy)-2,16,17-trihydroxyhexacosanoic acid; Hexacosanoic acid, 21-((O-6-O-acetyl-beta-D-glucopyranosyl-(1-2)-O-beta-D-xylopyranosyl-(1-2)-4-O-acetyl-beta-D-xylopyranosyl)oxy)-2,16,17-trihydroxy-, (2S-(2R*,16S*,17R*,21S*))- |
| IUPAC Name | (2S,16R,17S,21R)-21-[(2S,3R,4S,5R)-5-acetyloxy-4-[(2S,3R,4S,5S,6R)-6-(acetyloxymethyl)-3,4,5-trihydroxyoxan-2-yl]oxy-3-[(2S,3R,4S,5R)-3,4,5-trihydroxyoxan-2-yl]oxyoxan-2-yl]oxy-2,16,17-trihydroxyhexacosanoic acid |
| Canonical SMILES | CCCCCC(CCCC(C(CCCCCCCCCCCCCC(C(=O)O)O)O)O)OC1C(C(C(CO1)OC(=O)C)OC2C(C(C(C(O2)COC(=O)C)O)O)O)OC3C(C(C(CO3)O)O)O |
| InChI | InChI=1S/C46H82O21/c1-4-5-15-19-29(20-18-23-31(50)30(49)21-16-13-11-9-7-6-8-10-12-14-17-22-32(51)43(58)59)64-46-42(67-44-39(56)36(53)33(52)24-61-44)41(35(26-62-46)63-28(3)48)66-45-40(57)38(55)37(54)34(65-45)25-60-27(2)47/h29-42,44-46,49-57H,4-26H2,1-3H3,(H,58,59)/t29-,30-,31+,32+,33-,34-,35-,36+,37-,38+,39-,40-,41+,42-,44+,45+,46+/m1/s1 |
| InChI Key | UBCWOWKILIETKQ-NSMKLYECSA-N |
Properties
| Appearance | Amorphous Powder |
| Antibiotic Activity Spectrum | Gram-positive bacteria |
| Boiling Point | 1049.1±65.0 °C (Predicted) |
| Density | 1.31±0.1 g/cm3 (Predicted) |
Reference Reading
1. Structure elucidation of glykenin glycosidic antibiotics from Basidiomycetes sp. V. High-performance liquid chromatographic separation of components of glykenin
F Nishida, M Nishimura, K Harada, M Suzuki, V Meevootisom, T W Flegel, Y Thebtaranonth, S Intararuangsorn J Chromatogr A. 1994 Apr 1;664(2):195-202. doi: 10.1016/0021-9673(94)87008-x.
The glycosidic antibiotics of the glykenin (GK) family produced by Basidiomycetes sp. were separated into nine components (GK-I-VII and DG) by normal-phase chromatography. It was found that these components differ in the number and location of the acetyl groups in the sugar moiety. Each component (GK-I-VII and DG) was further separated into three isomers (A, B and C), which possess different aglycones, by reversed-phase chromatography on an ODS column with methanol-acetonitrile as eluent. The best composition of the eluent was found to be methanol-acetonitrile-1% trifluoroacetic acid (4:3.5:2.5). The profile analysis of GK-III-VII and DG was also carried out using a modified mobile phase. The combination of normal- and reversed-phase chromatography separated all components of the GK mixture except GK-I and II. The relationship between structure and separation behaviour of GK is discussed.
Recommended Products
| BBF-03753 | Baicalin | Inquiry |
| BBF-03754 | CASTANOSPERMINE | Inquiry |
| BBF-00764 | Cerebroside C | Inquiry |
| BBF-00677 | 3-Amino-3-deoxy-D-glucose | Inquiry |
| BBF-05862 | Epirubicin | Inquiry |
| BBF-01825 | Loganin | Inquiry |
Bio Calculators
* Our calculator is based on the following equation:
Concentration (start) x Volume (start) = Concentration (final) x Volume (final)
It is commonly abbreviated as: C1V1 = C2V2
* Total Molecular Weight:
g/mol
Tip: Chemical formula is case sensitive. C22H30N4O √ c22h30n40 ╳