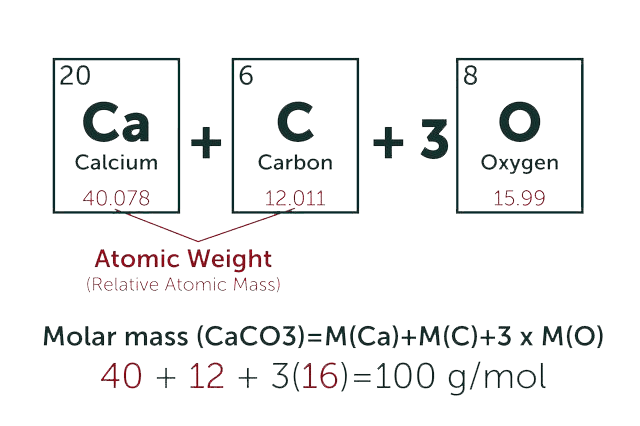Sulfurmycin F
* Please be kindly noted products are not for therapeutic use. We do not sell to patients.
| Category | Antibiotics |
| Catalog number | BBF-03094 |
| CAS | 83829-33-8 |
| Molecular Weight | 841.89 |
| Molecular Formula | C43H55NO16 |
Online Inquiry
Description
Sulfurmycin F is an anthracycline antibiotic produced by Str. galilaeus OBB-111. It has anti-Gram-positive bacteria and anti-tumor activity.
Specification
| Synonyms | 1-Naphthacenecarboxylic acid, 1,2,3,4,6,11-hexahydro-2,5,7-trihydroxy-6,11-dioxo-2-(2-oxopropyl)-4-((2,3,6-trideoxy-4-O-(2,6-dideoxy-4-O-((2S-(2alpha,5beta,6beta))-tetrahydro-5-hydroxy-6-methyl-2H-pyran-2-yl)-alpha-L-lyxo-hexopyranosyl)-3-(dimethylamino)-alpha-L-lyxo-hexopyranosyl)oxy)-, methyl ester, (1R-(1alpha,2beta,4beta))- |
| IUPAC Name | methyl (1R,2S,4S)-4-[(2R,4S,5S,6S)-4-(dimethylamino)-5-[(2S,4S,5S,6S)-4-hydroxy-5-[(2S,5S,6S)-5-hydroxy-6-methyloxan-2-yl]oxy-6-methyloxan-2-yl]oxy-6-methyloxan-2-yl]oxy-2,5,7-trihydroxy-6,11-dioxo-2-(2-oxopropyl)-3,4-dihydro-1H-tetracene-1-carboxylate |
| Canonical SMILES | CC1C(CCC(O1)OC2C(OC(CC2O)OC3C(OC(CC3N(C)C)OC4CC(C(C5=CC6=C(C(=C45)O)C(=O)C7=C(C6=O)C=CC=C7O)C(=O)OC)(CC(=O)C)O)C)C)O |
| InChI | InChI=1S/C43H55NO16/c1-18(45)16-43(53)17-29(34-23(36(43)42(52)54-7)13-24-35(39(34)51)38(50)33-22(37(24)49)9-8-10-27(33)47)58-31-14-25(44(5)6)40(20(3)56-31)60-32-15-28(48)41(21(4)57-32)59-30-12-11-26(46)19(2)55-30/h8-10,13,19-21,25-26,28-32,36,40-41,46-48,51,53H,11-12,14-17H2,1-7H3/t19-,20-,21-,25-,26-,28-,29-,30-,31-,32-,36-,40+,41+,43+/m0/s1 |
| InChI Key | ONRDFNTYASFICP-QVSAPZMJSA-N |
Properties
| Antibiotic Activity Spectrum | Gram-positive bacteria; neoplastics (Tumor) |
| Boiling Point | 933.2±65.0°C at 760 mmHg |
| Melting Point | 151.5°C |
| Density | 1.4±0.1 g/cm3 |
Reference Reading
1. New anthracycline antibiotics, auramycins and sulfurmycins. II. Isolation and characterization of 10 minor components (C approximately G)
T Hoshino, M Tazoe, S Nomura, A Fujiwara J Antibiot (Tokyo). 1982 Oct;35(10):1271-9. doi: 10.7164/antibiotics.35.1271.
Following the discovery of new anthracycline antibiotics, auramycins A and B and sulfurmycins A and B, we found 10 minor analogues of auramycins and sulfurmycins, C, D, E, F and G, from the culture broth of a mutant strain of Streptomyces galilaeus OBB-111 and prepared 2 analogues as the chemical derivatives from auramycin G and sulfurmycin G. All analogues have a sugar moiety at C-7 position of the aglycones. These analogues exhibit activities against Gram-positive bacteria and P388 leukemia.
Recommended Products
| BBF-02642 | Lactonamycin | Inquiry |
| BBF-00764 | Cerebroside C | Inquiry |
| BBF-03754 | CASTANOSPERMINE | Inquiry |
| BBF-05862 | Epirubicin | Inquiry |
| BBF-03753 | Baicalin | Inquiry |
| BBF-00677 | 3-Amino-3-deoxy-D-glucose | Inquiry |
Bio Calculators
* Our calculator is based on the following equation:
Concentration (start) x Volume (start) = Concentration (final) x Volume (final)
It is commonly abbreviated as: C1V1 = C2V2
* Total Molecular Weight:
g/mol
Tip: Chemical formula is case sensitive. C22H30N4O √ c22h30n40 ╳