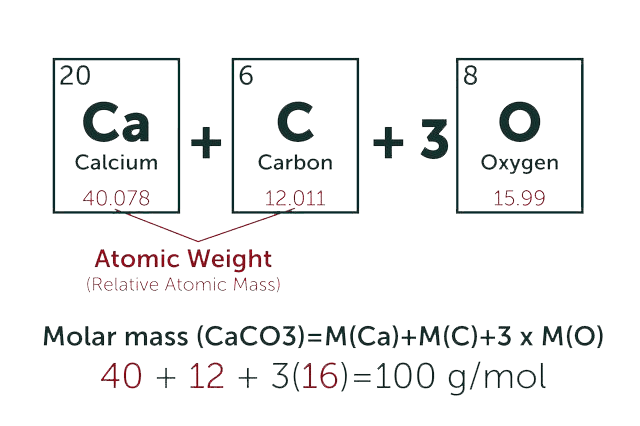Demethylchromomycin A3
* Please be kindly noted products are not for therapeutic use. We do not sell to patients.
| Category | Antibiotics |
| Catalog number | BBF-00813 |
| CAS | 86917-64-8 |
| Molecular Weight | 1169.22 |
| Molecular Formula | C56H80O26 |
Online Inquiry
Description
Demethylchromomycin A3 is an antitumor antibiotic produced by Streptomyces aburaviensis. It has anti-Gram-positive bacteria and anti-leukemia P388 cell activity.
Specification
| Synonyms | Olivomycin D, 3D-(4-O-acetyl-2,6-dideoxy-3-C-methyl-alpha-L-arabino-hexopyranosyl)-4B-demethyl-7-methyl- |
| IUPAC Name | [6-[[6-[4-[4-(5-acetyloxy-4-hydroxy-4,6-dimethyloxan-2-yl)oxy-5-hydroxy-6-methyloxan-2-yl]oxy-5-hydroxy-6-methyloxan-2-yl]oxy-7-(3,4-dihydroxy-1-methoxy-2-oxopentyl)-4,10-dihydroxy-3-methyl-5-oxo-7,8-dihydro-6H-anthracen-2-yl]oxy]-4-(4,5-dihydroxy-6-methyloxan-2-yl)oxy-2-methyloxan-3-yl] acetate |
| Canonical SMILES | CC1C(C(CC(O1)OC2CC(OC(C2OC(=O)C)C)OC3=CC4=CC5=C(C(=O)C(C(C5)C(C(=O)C(C(C)O)O)OC)OC6CC(C(C(O6)C)O)OC7CC(C(C(O7)C)O)OC8CC(C(C(O8)C)OC(=O)C)(C)O)C(=C4C(=C3C)O)O)O)O |
| InChI | InChI=1S/C56H80O26/c1-20-33(78-39-18-36(52(25(6)74-39)76-27(8)58)81-37-15-32(60)46(63)22(3)71-37)14-30-12-29-13-31(53(70-11)51(68)45(62)21(2)57)54(50(67)43(29)49(66)42(30)44(20)61)82-40-17-34(47(64)24(5)73-40)79-38-16-35(48(65)23(4)72-38)80-41-19-56(10,69)55(26(7)75-41)77-28(9)59/h12,14,21-26,31-32,34-41,45-48,52-55,57,60-66,69H,13,15-19H2,1-11H3 |
| InChI Key | LKAAYCHLHTYRFV-UHFFFAOYSA-N |
Properties
| Appearance | Orange Powder |
Reference Reading
1. NMR studies of chromomycins, olivomycins, and their derivatives
Y Yoshimura, M Koenuma, K Matsumoto, K Tori, Y Terui J Antibiot (Tokyo). 1988 Jan;41(1):53-67. doi: 10.7164/antibiotics.41.53.
Detailed studies on the 13C and 1H NMR spectra of chromomycins A2 and A3, olivomycins A and B, and their derivatives clarified the assignment of many signals which had been unassigned or erroneously reported in the literatures. The revised assignments for chromomycin A3 and olivomycin A include the assignment of a key 13C signal used to discuss the saccharide linkage in question. Structure analyses based on the revised assignments support the alpha,1----3-bond between components of the disaccharide moiety in the molecules. Some general information useful for structure analysis of saccharides is also reported.
2. New aureolic acid antibiotics. II. Structure determination
M Koenuma, Y Yoshimura, K Matsumoto, Y Terui J Antibiot (Tokyo). 1988 Jan;41(1):68-72. doi: 10.7164/antibiotics.41.68.
Structure determination using NMR spectroscopy of new aureolic acid analogues, demethylchromomycins A2 and A3 and demethylolivomycins A and B produced by Streptomyces aburaviensis PA-39856, is described.
Recommended Products
| BBF-00677 | 3-Amino-3-deoxy-D-glucose | Inquiry |
| BBF-02614 | Nystatin | Inquiry |
| BBF-01693 | Doxorubicin EP Impurity A (Daunorubicin) | Inquiry |
| BBF-05862 | Epirubicin | Inquiry |
| BBF-03755 | Actinomycin D | Inquiry |
| BBF-01829 | Deoxynojirimycin | Inquiry |
Bio Calculators
* Our calculator is based on the following equation:
Concentration (start) x Volume (start) = Concentration (final) x Volume (final)
It is commonly abbreviated as: C1V1 = C2V2
* Total Molecular Weight:
g/mol
Tip: Chemical formula is case sensitive. C22H30N4O √ c22h30n40 ╳